- Pick up from the Samtech Store in Ambala
To pick up today
Free
- Courier delivery
Our courier will deliver to the specified address
4-5 Days
200+

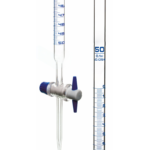
₹200 Original price was: ₹200.₹90Current price is: ₹90. (Exc. GST)
To pick up today
Free
Our courier will deliver to the specified address
4-5 Days
200+
Performing a platinum wire test, also known as the “flame test,” is a common experiment in chemistry labs, especially for students learning about the properties of different metal ions. The test involves heating a small loop of platinum wire in a Bunsen burner flame and observing the color produced when different metal ions are introduced. This is a qualitative analysis method used to identify the presence of specific metal ions based on the characteristic colors they emit. It is crucial for students to adhere to safety guidelines when working with open flames and hot materials in the laboratory. Additionally, proper disposal of the contaminated wire and chemicals used in the test is essential to avoid contamination and potential hazards.
When conducting the platinum wire test, students should first clean the platinum wire loop to ensure that any residue from previous experiments is removed. Then, the cleaned wire is dipped into the sample of the compound to be tested and placed in the Bunsen burner flame for observation. The flame color is compared with known reference colors to identify the metal ion present. This experiment not only provides hands-on experience with laboratory techniques but also helps students understand the unique emission spectra of different elements. It is essential for students to conduct this experiment under the supervision of a knowledgeable instructor to ensure proper technique and safety measures are followed throughout the process.
In stock
In stock
In stock
In stock
In stock
No account yet?
Create an Account
SAMTECH INSTRUMENTS
Typically replies within minutes
Any questions related to Platinum Loop?
🟢 Online | Privacy policy
WhatsApp us

